Revolutionizing Cerebral Endovascular Therapy with the World’s First Polymer Membrane-based Flow Diverter
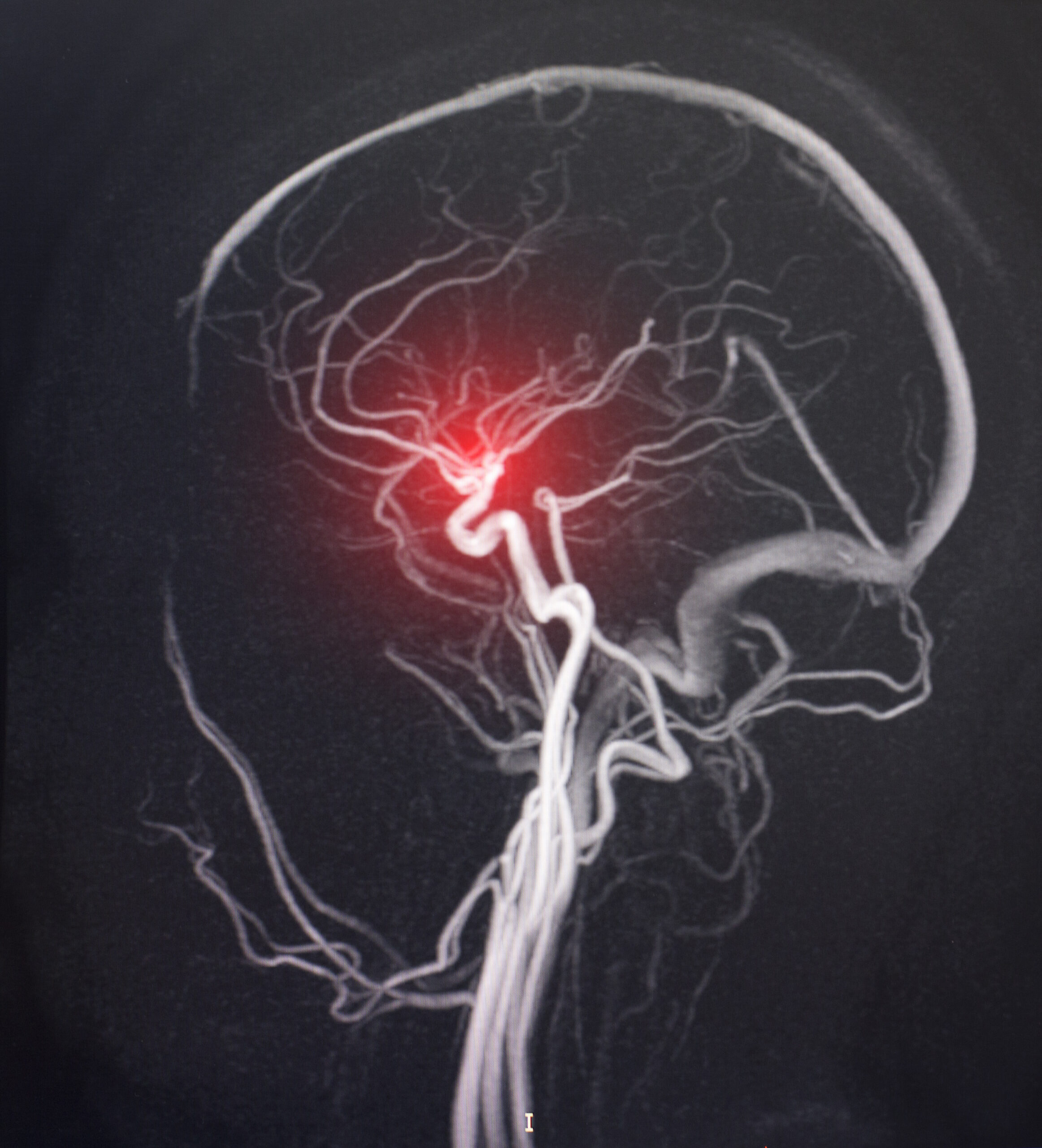
Our flow-diversion technology is based on a microporous polymer membrane that is delivered to the lesion location using a laser cut implant-frame. The laser drilled membrane can be designed to provide a surface coverage ranging from 10% to 100%, to achieve an optimal performance.
KEY BENEFITS
- Efficient flow-diversion leading to single device treatment
- Precise positioning with expandable balloon for less foreshortening and better flow diversion
- Superior wall appositioning with laser-cut frame
- Atraumatic implant surface with minimal metal interface
- Customizable porosity with option to vary coverage along the length
- Potential to lessen anti-platelet pharmacotherapy requirement
- Higher coverage of aneurysm occlusion results in lower risk of late-stage aneurysm ruptures
INTELLECTUAL PROPERTY
The technology behind Merlin’s family of products is supported by a portfolio of granted patents in:
- USA
- Europe
- Japan
KEY APPLICATIONS
Multipurpose technology platform
For Haemorrhagic applications, the membrane provides an efficient flow-diversion while providing an atraumatic scaffold for the endothelial layer regrowth. The same membrane can also be deployed to catch and retrieve a clot in Ischemic applications.
Highly versatile and effective
- Our devices work with all standard auxiliary devices
- Our devices offer 50-75% coverage (vs. <30% for most others)
- Our devices have far lesser metal content
- Made in Singapore for the best precision and quality assurance

